Manufacturing: Tabletting
Critical Tablet Press Design Considerations for Continuous Manufacturing Applications
What are the differences and key design features which characterise successful tablet press performance in a continuous manufacturing setting?
By Frederick Murray at KORSCH
Once considered to be the driver of a dramatic paradigm shift in the production methodology for pharmaceutical tablet production, continuous manufacturing technology is still gaining traction as formal FDA approval is granted for several new products. Today, batch production remains the standard for the vast majority of the industry.
The advantages of continuous manufacturing can be considerable, from reduced API consumption during product development, to the extensive use of PAT tools to support immediate release, a reduced GMP space requirement and increased safety in the manufacture of highly potent products. Still, equipment capital costs and formidable validation challenges have dampened the widespread adoption of continuous technology.
The tablet press – a key component in both batch and continuous manufacturing – is continuous by nature. The press will run as long as the material level sensor detects product. For most advanced tablet presses, a highly efficient dust extraction system, when paired with an equally effective punch lubrication system, will ensure a clean compression zone and the ability of the tablet press to run continuously for many hours – or even days – with minimal intervention.
For large campaign batch production, it is not uncommon for a high-speed tablet press to run for multiple weeks with just minor cleaning between successive batches.

Figure 1: Turret
Features Highlighting Continuous Manufacturing Compared to Batch Mode
For optimal continuous manufacturing line performance, there are several important features that differentiate the tablet press design from the batch mode of operation. Most continuous applications (5-20 kg/hour) do not require a large tablet press, but tablet size and the line production rate can require significant flexibility in a single machine. Per the example below (Table 1), a single press – with turret configurations of 16-station (TSM B, max tablet diameter of 16 mm) and 37-station (TSM BBS, max tablet diameter of 11 mm) – can accommodate a 5-20 kg line producing between 16,667 tablets per hour (300 mg each) and 200,000 tablets per hour (100 mg each), all within a comfortable machine operating speed.
Given the capital investment in the line, and the degree of automation integration between the press and the central supervisory system, a single press with a wide range of turret options is a key consideration for any continuous application (Figure 1).
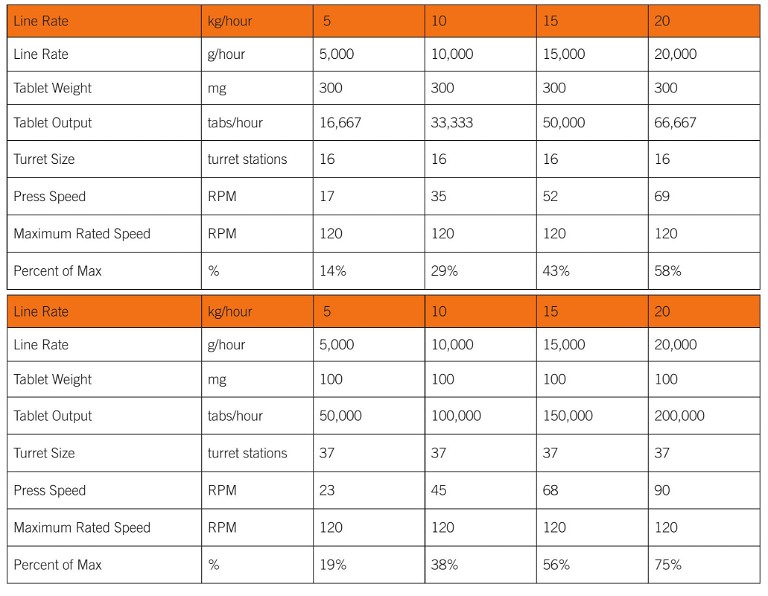
Table 1
'... equipment capital costs and formidable validation challenges have dampened the widespread adoption of continuous technology'

Figure 2: Gerricke Feeder
Every continuous production application requires the mechanical integration of the continuous feeding and blending system with the tablet press inlet hopper. This requires a high degree of flexibility in the mechanical design of the press hopper, which must mate cleanly to the overhead feeding system. For high potency applications, the press must be configured with the mechanical interface to the containment valve, which will feed material to the press in a contained manner (Figure 2).
To accommodate the in-process measurement of content uniformity, the tablet press in a continuous line must be able to support the placement of one or more NIR probes in the feeding pipe and, often, in the feeder itself. In most cases, the NIR probe is mounted through an existing sight glass in the feeder, with the feeder paddles modified to ensure the probe is accessing the material bed (Figure 3).
With precise loss in weight feeders and continuous blenders, it is important that homogeneity of the granulation is preserved as it moves through the tablet press feed pipe and feed frame. Some advanced tablet presses are configured with damping systems that eliminate vibration transmitted to the feeding system – an occurrence that can cause particle size segregation in the feeding system.
A key objective of continuous manufacturing is to match the rate of tablet production to the feed rate – a requirement that does not exist in batch production. In general, the system requires an analogue level sensor to precisely measure the column height of the material in the feeding system. Speed is then controlled by the press – in a close-loop fashion – to maintain material levels. A constant level means tablets are being produced at exactly the same rate the material is being fed to the tablet press. In batch production, a binary level sensor is utilised to confirm the presence of material and there is no closed loop control of the press speed.
A key feature common to both continuous and batch production is the automatic tablet weight control algorithm, commonly referred to as a press control system. Even with the lower outputs associated with continuous production, technology to measure individual tablet weights as fast as tablets can be produced does not exist.
In lieu of this, the tablet press monitors a secondary parameter – the compression force of each tablet – to confirm tablet weight. This force control system works as follows:

Figure 3: Sentronic Probe in the Feeder
- Tablets are produced by filling each die to a certain height as the dies pass under the feeder. The material in the die is then compressed between the upper and lower punch at the compression rollers, each to a uniform thickness
- When the material is compressed between the punches, a compression force is generated and measured at the compression rollers. This permits the measurement of the force associated with every tablet that is produced. When the target weight (die fill) and thickness are achieved, the press force associated with this tablet is then established as the process setpoint
- If all tablets are produced with the same amount of material in the die – and then compressed to the same thickness – the measured press force for each tablet should be the same
- Since the distance between the compression rollers – which determines the tablet thickness (space between the upper and lower punch tips) is fixed – it is only the amount of material in the die that can cause the compression force to vary, since the amount of material in the die determines tablet weight
- With this relationship between press force and tablet weight established – and with the ability to measure each and every press force as tablets are produced – the control system can measure the press forces in real time, calculate a moving average and make precise adjustments to the dosing cam (which controls the fill in the dies) to maintain the target tablet weight.
'A key objective of continuous manufacturing is to match the rate of tablet production to the feed rate – a requirement that does not exist in batch production'
The relationship between press force and tablet weight can also be used to develop stop limits; ensuring the press will stop before any bad tablets are produced. Most modern tablet presses offer a tablet rejection system which, on the basis of press force, can reject out of spec tablets as they are produced. The most advanced systems can reject a single tablet, at any press speed, and create a corresponding record to track the date, time and punch station that created the reject. Additional limits permit the press to stop based on excessive rejections, a high frequency of rejects over a short time or a preset number of rejects from the same punch station. In this way, the force control system constitutes a highly effective process analytical technology that monitors quality in real time with the ability to reject and report also in real time. Truly continuous production demands a well-designed and highly effective dust collection system; one that pulls dust from key areas around the turret to keep compression zones clean and production yields high. Some advanced presses offer a motorised damper tied to a negative pressure sensor – this permits closed loop control of the optimal dust extraction pressure. Similarly, a robust, automatic punch lubrication system is a prerequisite for continuous manufacturing. Over long-term operation, the punches must be lubricated. However, if excessive lubrication accumulates, the potential for tablet contamination is high. Some advanced tablet presses offer multi-zone lubrication systems, which offer the flexibility to control lubrication frequency and volume independently to the upper punch heads, upper punch barrels, lower punch heads and lower punch barrels. This flexibility allows the punch lubrication system to be optimised based on press speed, tool configuration (round or shaped) and compression force.
For tablet presses, perhaps the most significant difference between conventional batch mode and continuous process mode is control system configuration. In batch mode, the most advanced tablet presses are generally integrated to a central network for centralised batch data storage. Most modern machines have control systems that govern the operation of both the tablet press and its peripherals, including dedusting, metal check and, in some cases, an integrated tablet tester that measures tablet weight, thickness and hardness in real time, with closed loop control to the press force control system.
'For tablet presses, perhaps the most significant difference between conventional batch mode and continuous process mode is control system configuration'
In a continuous production application, the complete line is managed by a fully integrated supervisory control and data acquisition (SCADA) system. All press parameters are visible to the SCADA system and it controls the tablet press based on the PAT technology installed on the line. For example, the SCADA system may direct all tablets to the reject channel of the discharge chute based on the NIR measurements in the feed pipe or feed frame. The SCADA system can also stop the press if there is an issue with upstream or downstream process equipment. The complete product recipe is downloaded from SCADA system to press, ensuring the entire line is harmonised regarding starting process parameters and quality metrics. The SCADA system will monitor every fault, every adjustment and every tablet reject, and if required, will override the tablet press control system to make in-process adjustments. This advanced integration with the SCADA system for continuous manufacturing demands a highly flexible tablet press control system and a hardware/software architecture that can be integrated without major customisation. The fundamental controls of the tablet press remain intact, but the press is now operating as part of a fully integrated system – and the control system must support this mode of operation.
In summary, while the tablet press is inherently capable of continuous processing, there are a range of important design considerations required to ensure success in a continuous manufacturing setting. These features incorporate both mechanical and control system enhancements that permit seamless integration and full supervisory control. The most advanced tablet compression technology has a modular design that permits the same machine to be converted from batch to continuous mode, which is certainly an important consideration for any company evaluating a move toward a continuous production strategy.

Frederick Murray is CEO of KORSCH America, based in Boston, US, and the chief business development officer for KORSCH AG, based in Berlin, Germany. Mr Murray has been with KORSCH for 35 years, and during that time has authored and published technical papers on tablet press automation, tablet press validation, multi-tip tooling and process optimisation strategies. He has also taught courses on concepts of tablet compression, tablet press automation, multi-layer technology, tablet press containment and tablet press troubleshooting.