Advanced Contamination Detection Systems
Advanced contamination detection systems in GMP manufacturing
Miriam Krechlok from Mettler-Toledo Product Inspection speaks to IPT about advanced contamination detection systems and how these are utilised in pharma manufacturing to ensure seamless and good manufacturing practice-compliant products
IPT: What are the benefits and drawbacks of using X-ray to detect contaminants?
Miriam Krechlok (MK): Historically, contamination detection in pharma manufacturing relied heavily on manual visual inspection. Trained operators would physically examine products, containers or packaging to identify visible foreign materials or defects. This process formed an important early layer of quality assurance, particularly when production volumes were lower and packaging formats were less complex.
However, visual inspection presented clear limitations. It was time-intensive, dependent on human concentration, and subject to variability between operators and shifts. Small contaminants, low-contrast foreign bodies or particles concealed within packaging could easily be missed. As pharma manufacturing scaled and regulatory scrutiny increased, the need for more reliable and repeatable detection methods became evident.
IPT: What technologies are currently used for physical contamination detection?
MK: Today, physical contamination detection in pharma production is primarily conducted using metal detection and x-ray inspection technologies. Metal detectors are highly effective at identifying ferrous and non-ferrous metals plus stainless steel contaminants within bulk product or finished packs. It is commonly implemented during early processing stages, such as powder handling or tabletting, where metallic fragments may originate from sieves, tooling or processing equipment. X-ray inspection systems extend this capability further by detecting a broader range of foreign bodies including glass, mineral stone and dense plastics. In addition, X-ray technology can simultaneously verify product presence, fill level and pack completeness through sealed primary and secondary packaging. The selection of inspection technology should be based on a documented risk assessment considering product characteristics, manufacturing process, production speed and packaging configuration. Performance, detection sensitivity and rejection mechanisms must be verified as part of equipment qualification and ongoing performance monitoring.
IPT: How were contaminants historically detected during pharma manufacturing?
MK: X-ray inspection provides one of the most comprehensive methods for detecting physical contamination in pharma manufacturing. Its primary advantage lies in its versatility: X-ray systems can identify a wide range of foreign bodies including glass, mineral fragments and certain dense plastics. This broader detection capability makes X-ray particularly valuable in environments where contamination risks are varied or difficult to predict.
Another significant benefit is the ability to inspect products through packaging. X-ray systems can operate effectively after products have been sealed in blister packs, bottles or cartons, even when metallised films or foils are present. In terms of X-ray drawbacks, in vertical form, fill and seal applications, metal detection technology is the only viable product inspection solution. Before the tabletting process, powdered ingredients can be inspected for ferrous and non- ferrous metals plus stainless steel contaminants. Such foreign bodies can result from broken sieves or processing equipment. Contaminated product is detected and removed from production before it can be further broken down or additional valuable time and product is added.

The implementation of contamination detection systems must be justified through a risk-based evaluation and integrated at risk-based inspection points or in-process control steps. In cases where risk assessment demonstrates that metallic contamination represents the primary and reasonably foreseeable hazard, metal detection may be sufficient and operationally appropriate.
IPT: Why is secure and defect-free packaging important in the pharma sector?
MK: Secure and defect-free packaging is fundamental to pharma product safety, stability and regulatory compliance. Packaging serves as the primary barrier protecting medicines from environmental exposure, contamination and physical damage throughout distribution and storage. Any compromise to that barrier can directly affect product efficacy and patient safety. Defects such as missing or broken products and packaging and/or label errors can compromise product stability, sterility assurance and patient safety. For sterile or high sensitivity products, even minor packaging failures may result in a batch rejection. Packaging also plays a critical role in traceability and regulatory communication. Labels must display accurate batch numbers, expiry dates, dosage information and compliance markings. Errors in labelling or print quality can lead to misidentification, dispensing errors or regulatory non-compliance. For these reasons, packaging inspection is closely integrated into pharma quality control strategies. Technologies such as vision inspection and X-ray inspection help verify pack integrity, component presence and seal quality. When validated and integrated within the pharma quality system, these systems contribute to documented evidence of process control and compliance with good manufacturing practice (GMP) requirements.
In such a tightly regulated environment, accuracy underpins both compliance and safety. GMP and the wider good x practice framework require that products are consistently produced and controlled according to defined quality standards. Dynamic weighing supports these requirements by confirming that each unit meets its declared weight and content specifications.
A precision checkweighing system helps to:
• Demonstrate regulatory compliance through verified data for validation and qualification records such as design qualification, installation, operational and performance qualification
• Reduce quality costs by lowering the risk of manual error, rework and waste
• Protect patient safety through consistent quality that reinforces confidence among healthcare professionals and the end recipient.
IPT: How is automation being integrated into pharma manufacturing to streamline processes?
MK: Automation is deeply embedded within modern pharma manufacturing, spanning formulation, filling, packaging and labelling processes. Increasingly, this automation is extending into quality control and contamination detection, enabling manufacturers to maintain high production throughput while upholding strict safety and compliance standards. Automated inspection systems such as metal detectors and X-ray units operate continuously at line speed, inspecting all product output rather than relying on manual sampling. This improves detection consistency while reducing the operational burden on quality teams. Automated reject mechanisms remove contaminated or defective products in real time, preventing them from progressing further through the production process.
Another example of successful automation is the smart fill level adjustment feature on checkweighers that uses dynamic feedback to automatically regulate filling equipment in real time. Rather than relying on manual checks or occasional adjustments, the smart fill level adjustment feature on the checkweigher monitors the average weight of a series of packs and instructs upstream filling machines and multi-head weighers to make micro-corrections. The result is a tighter weight distribution, more consistent pack quality and regulatory compliance. To meet regulatory expectations, electronic records must comply with applicable data integrity requirements. Features such as secure user management, audit trails, controlled data storage and restricted access are essential to allow manufacturers to have the confidence that inspection data can be relied upon during audits, investigations and batch certification. Integration with data management platforms enables centralised oversight across multiple inspection systems, supporting trending, deviation analysis and continuous process verification.

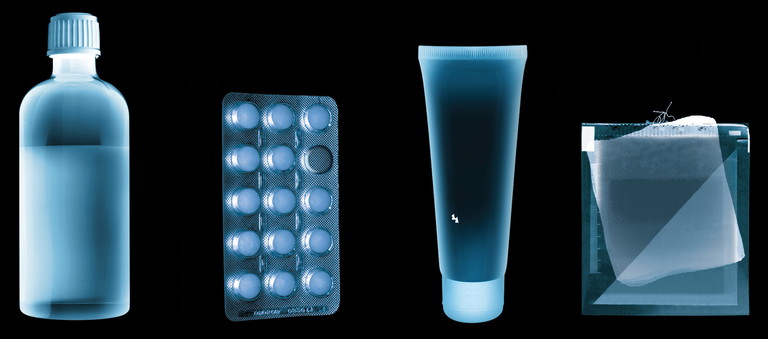
Automation also supports process optimisation. Integrated inspection and data management systems can communicate with upstream and downstream equipment, enabling coordinated responses to quality deviations. For example, recurring contamination events may trigger alerts, production adjustments or preventative maintenance actions. By combining automated inspection, digital documentation and connected system intelligence, pharma manufacturers can streamline operations while strengthening compliance, traceability and product protection.
IPT: How will advanced contamination detection systems develop over the next five years?
MK: Over the next five years, advanced contamination detection systems are expected to evolve in both technical capability and operational integration. One of the most significant developments will be increased detection sensitivity. Continued refinement of detection algorithms incorporating artificial intelligence will enhance the performance of both metal detection and X-ray technologies, enabling them to further improve product safety and integrity checks. Alongside sensitivity improvements, connectivity will play an increasingly important role. Inspection systems will become more tightly integrated with manufacturing execution systems and wider production networks, which will enable real-time data exchange, faster operator notifications and more coordinated responses to the detection of faulty products. Automation of documentation will also advance. Inspection activities, test routines and performance verification will be recorded automatically, creating comprehensive digital audit trails. This will simplify compliance reporting while providing manufacturers with deeper operational insight.
While distinct from physical contamination detection, microbial testing technologies are also progressing, particularly in rapid detection and analysis methods. Together, these innovations point towards a more connected, intelligent and preventative approach to pharma contamination control.

Miriam Krechlok is a segment marketing manager for Mettler-Toledo Product Inspection, based in Germany. Miriam is responsible for the food and pharma sectors, providing expertise and insights for business development and segmented marketing campaigns. Miriam’s background has seen her work in corporate identity and marketing in the food, cosmetics and pharma industries. Her previous role at Mettler-Toledo was head of marketing for the DACH region.