Fluorescence-Guided Surgery
From mouse to medicine: the future of NIR-II FGS
As near-infrared imaging advances beyond the NIR-I window, a new generation of NIR-II fluorescent probes is redefining intraoperative visualisation. With improved tissue penetration, reduced scattering and enhanced signal-to-background ratios, these next-generation agents are expanding in vivo applications and opening new perspectives for precision surgery
Romain Membrat and Olivier De Beaumont at Provepharm
Fluorescence-guided surgery (FGS) is a cutting-edge medical imaging technique aimed at assisting highly complex surgical manoeuvres.1 The mainstream of minimally invasive surgical procedures such as laparoscopy, celioscopy or robot-assisted surgery has led to a progressive loss of surgeons’ ability to perceive tactile feedback. In this context, extensive efforts have been devoted to the development of paired fluorescence cameras and fluorescent probes to provide greater precision during intraoperative navigation.
Despite US Food and Drug Administration (FDA) approval of several fluorescent probes – indocyanine green (ICG) 1959, fluoresceine 1960, 5-ala 2017 and pegulicianine 2024 – ICG remains the gold standard. Owing to its availability, suitable fluorescence brightness and highly favourable toxicity profile, ICG is widely used in FGS today.2 However, achieving increasingly precise and well-defined images remains challenging. More recently, the need to associate quantitative figures with fluorescence images has emerged as an essential feature to standardising medical decision-making.
In this context, the scientific research in FGS is driven by two major objectives: the design of smart fluorescent probes (able to highlight specific targets); and the optimisation of the spectroscopic performance of the camera/fluorescent probe system.
The basic principle of fluorescence cameras relies on the device’s ability to excite fluorescent probes with an excitation beam, and to collect the resulting emission signal within a well-selected wavelength range. Currently approved ICG-compatible cameras are designed to collect emission signals in the NIR-I region of the optical spectrum (800-900nm for ICG).1,2
The spectroscopic limitations of this window have led researchers to show increasing interest in fluorophores operating in the NIR-II region, between 1,000-3,000nm.3 Due to lower biological tissue autofluorescence and reduced light scattering at these wavelengths, the penetration depth of the NIR-II fluorescence signal is significantly increased – approximately 1-2cm for NIR-Iand greater than 3cm for NIR-II. Consequently, NIR-II enables deeper visualisation of structures and results in lower image broadening, improved signal-to-noise ratio (SNR) and higher spatial resolution (up to 30µm at 1-3cm deep).4 Recent literature provides numerous comparisons between NIR-I and NIR-II, where NIR-Iimages are often linked to naked-eye observation, whereas NIR-II images correspond to the enhanced view provided by corrective lenses (Figure 1).5
Despite the tremendous advantages afforded by NIR-II fluorescence technique, only a few clinical examples of NIR-II FGS on humans have been published to date. The absence of an FDA-approved NIR-II-compatible fluorescent probe on the market provides a plausible explanation for the sluggish translation from fundamental research to routine clinical applications.6
The new generation of NIR-II fluorescent probes
The maturation of NIR-II FGS technologies requires the synergistic development of both fluorescent probes and spectro-optical cameras technologies. ICG is not a suitable fluorophore for the NIR-II window. The maximum emission of ICG is centred around 830nm, corresponding only to the tail of the NIR-II emission bandwidth (greater than 1,000nm).7 This results in low fluorescence intensity and rapid photobleaching. Small molecule-based fluorophores offer the advantage of being derived from well-known and biocompatible structures (such as ICG), with rapid clearance and well-characterised metabolism.8 To shift the emission spectrum towards the NIR-II region (redshift), several methodologies have been implemented based on chemical modification of a π-conjugation system to achieve higher emission wavelengths.9 Despite the recent publication of extensive work in this field, organic NIR-II fluorescent probes suffer from several drawbacks, such as high sensitivity of this electron-rich molecule, to oxidation and reduced fluorescence brightness as the emission wavelength increases. To address these limitations, the emergence of inorganic nanoparticles-based fluorescence probes must be considered.10 In contrast to organic molecules, nanoparticles-based probes provide enhanced photostability and highly competitive fluorescence performance in the NIR-II window (eg, quantum dots, rare-earth nanoparticles, gold nanoclusters and single-walled carbon nanotubes). However, although inorganic nano-based fluorescent probes may offer a viable solution, their transition to clinical application is hindered by pharma compatibility issues, such as metabolic retention and long-term toxicity. Despite recent efforts devoted to hybrid organic/inorganic materials, the development of a probe combining high fluorescence performances with optimal biocompatibility remains an unresolved challenge.11
The new generation of NIR-II fluorescence camera
Switching emission collection from 700-900nm to 1,000nm and above constitutes a substantial technological leap. NIR-I fluorescence detection can be easily compared to a conventional visible-light camera equipped with a silicon charge-coupled device. This simple, compact and cost-effective system cannot be transposed to NIR-II imaging, where a specific detector made of indium, gallium and arsenide is required.12 This semiconductor alloy collects fluorescence emissions between 1,000-1,700nm. For higher wavelength ranges, more complex photosensitive semiconductors based on indium antimonide or mercury telluride are used.5
In addition to being very expensive, these semiconductor detectors generate self-induced dark current, resulting in undesired thermal noise. Therefore, the detector must be cooled to temperatures between -20 to -80°C to reduce SNR, thereby increasing costs and equipment dimensions.4,5 Handling an NIR-II fluorescence camera in the operating room presents challenges not encountered with NIR-I systems. To date, no NIR-II-compatible FGS camera has been approved by health authorities. However, when the imaging system is precisely calibrated, NIR-II technologies can yield tremendous improvement in image quality, as illustrated by several limited clinical applications (Figure 2).
In vivo applications of NIR-II fluorescence imaging: case-based demonstrations
The major benefits of NIR-II fluorescence versus NIR-Ibecome apparent only when considering the interaction between radiation and biological tissue. Consequently, only in vivo fluorescence imaging can provide appropriately illustrated assessments.
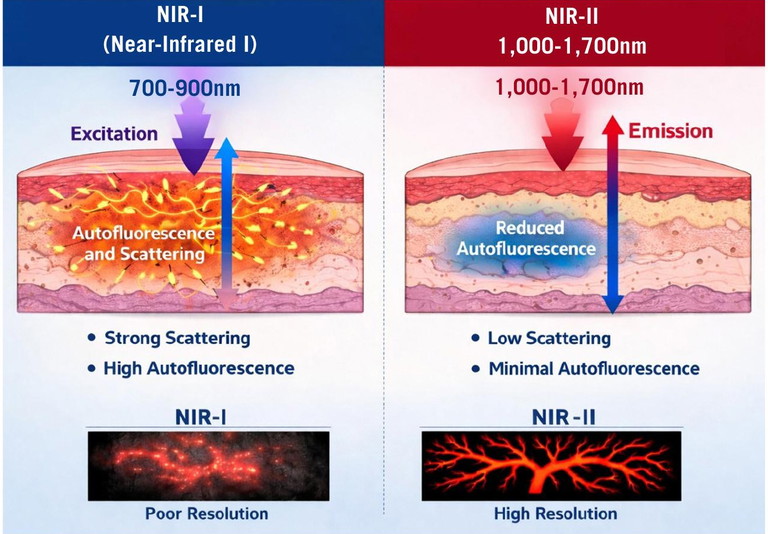
Figure 1 : Comparison of NIR-I versus NIR-II features
Preclinical cases
Preclinical NIR-II FGS studies have been mainly conducted on mice and rats, more sporadically on pigs and monkeys.4,5 The first example of in vivo application of NIR-II fluorescence was proposed in 2012 for visualisation of the vascular structure of a mouse’s hind limb with high spatial resolution.13 Since this pioneering work, numerous preclinical applications of NIR-II fluorescence have been published in a range of indications.5,10
Clinical applications
Because of the aforementioned reasons, the literature on clinical cases of NIR-II imaging remains very limited. Few clinical applications, all led by Chinese research groups, have been reported since 2019. The first-in-human application was published in 2019 by Hu et al, who used a homemade multispectral camera (NIR-I and NIR-II) and ICG as the fluorescent probe (0.5mg/kg).14
The authors compared NIR-I, NIR-II and white light (WL) for liver tumour delineation. In addition to improved quantitative performance (tumour-to-noise ratio 5.33 versus 1.45, and detection rate 56.41% versus 46.15%), NIR-II FGS enabled better intraoperative strategic decision-making.14 In one patient, tumour resection appeared nearly complete with NIR-I imaging, whereas NIR-II revealed residual fluorescence in the tumour margin. These observations led the surgeon to perform a second resection to eliminate residual tumour tissue, thereby decreasing the risk of recurrence.
Another clinical case in hepatic tumour surgery was published by Zhang et al in 2022.15 That same year, Shi et al presented a clinical case using NIR-II FGS for detection and resection of high-grade glioma.16 A homemade multispectral camera was used in a cohort of 15 patients with suspected newly diagnosed, untreated glioma. Resection was performed under WL or fluorescence assistance NIR-I/NIR-II after an injection of 1mg/kg of ICG. Superior performance of NIR-II compared with NIR-Iwas demonstrated – sensitivity 100.00% versus 97.40%; specificity 91.36% versus 90.91%; and precision 97.02% versus 91.91%.16 These differences are clinically relevant for highly complex and high-risk surgical procedures, such as neurosurgery.
A few months later, Cheng’s group published the first application of NIR-II imaging in microsurgery.17 Using ICG (0.5mg/kg) as the fluorescent probe, they used NIR-II fluorescence to assess the vascular integrity of anastomosis, vascularisation of a finger following digital reimplantation, prediction of necrosis after avulsion injury reconstruction and detection of skin perforators. Although the authors did not provide quantitative metrics associated with their surgical procedures, the NIR-II images gave better resolution, higher contrast and cleaner structural visualisation, thereby increasing the surgeon’s confidence. NIR-II FGS was also clinically applied in nephrological surgery for the resection of cystic renal masses using an ICG-derived fluorophore (IgGIRDye800CX).18
More recently, two clinical cases were published. In 2025, Zhang et al compared NIR-II and WL for orbital tumour surgery.19 Significantly higher sensitivity (80.0% versus 66.7%) and specificity (71.4% versus 42.8%) were reported, although no comparison was made with the use of ICG and NIR-I FGS.20 Dai et al recently reported the use of ICG and NIR-II FGS to guide oesophagectomy in a single patient. NIR-II provided highly contrasted and well-resolved visualisation to monitor oesophageal perfusion, whereas NIR-I fluorescence exhibited signal broadening.
Even if only a limited number of these studies presented quantitative data to demonstrate the superiority of NIR-II over NIR-I, all intraoperative images were highly convincing, showing improved contrast, higher resolution and more comfortable intraoperative navigation.
The future of NIR-II FGS
The rise of FGS has revolutionised operating rooms over the past fifteen years. Today, NIR-II technology could represent the next step. Despite the persistent challenges related to both cameras and fluorophores, the visual and quantitative improvement demonstrated in the few published studies are outstanding. The acceleration of NIR-II fluorescence adoption depends on compatible fluorophores emitting at longer wavelengths and on the design of NIR-II cameras adapted to minimally invasive surgery, including endoscopic or laparoscopic imaging instruments. Therefore, an ecosystem of complementary technologies – fluorophore pharmaceutical industries, camera manufacturers and artificial intelligence software developers – is necessary to foster new applications of dual imaging NIR-I and NIR-II fluorescence-guided surgery to address unmet medical needs in precision intraoperative navigation. It can reasonably be expected that once NIR-II imaging demonstrates substantial changes in clinical management, with significant improvements in patient survival, pharma companies and camera manufacturers will intensify their efforts to secure regulatory approval for new fluorophores and associated imaging systems. In this dynamic environment, scientific platforms are specifically designed to accelerate research, de-risk development and facilitate market access for next-generation fluorophores.
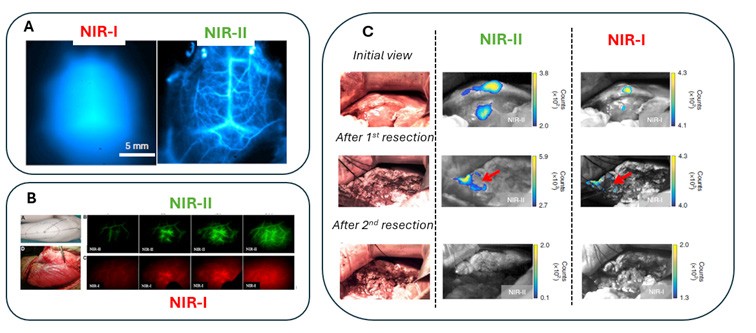
Figure 2 : Example of preclinical and clinical applications of NIR-II imaging technologies. (A) brain vascularisation mapping of a mouse viewed in NIR-I and NIR-II; B) visualisation of perforating vessel in reconstructive surgery on humans; (C) clinical comparison of liver tumour resection assisted by NIR-I and NIR-II imaging3,14,17
The high level of quality achieved with NIR-II imaging may ultimately pave the way for quantitative fluorescence, enabling systematic standardisation of the surgical decision-making.
References:
1. Ullah Z et al (2024), ‘Fluorescence imaging-guided surgery: current status and future directions’, Biomaterials Science, 12, 3765-3804
2. Franseva P (2024), ‘A Green Lantern for the Surgeon: A Review on the Use of Indocyanine Green (ICG) in Minimally Invasive Surgery’, J Clin Med, 13, 4895
3. Wang F (2024), ‘In vivo NIR-II fluorescence imaging for biology and medicine’, Nature Photonics,18, 535-547
4. Schmidt E L (2024), ‘Near-infrared II fluorescence imaging’, Nature Reviews Methods Primers, 4(23)
5. Li N (2024), ‘Progress of NIR-II fluorescence imaging technology applied to disease diagnosis and treatment’, Eur J Med Chem, 267, 116173
6. Lawrance R (2024), ‘The luminous frontier: transformative NIR-IIa fluorescent polymer dots for deep-tissue imaging’, RSC App Polym, 2, 749-774
7. Starosolski Z (2017), ‘Indocyanine green fluorescence in second near-infrared (NIR-II) window’, PLOS One, 9, 1-14
8. Guo M (2025), ‘Near-Infrared-II Fluorescence Imaging of Tumors with Organic Small-Molecule Fluorophores’, Sensors, 25, 7080
9. Zou M (2026), ‘Advances in the near-infrared II for in vivo fluorescence imaging applications: A review’, Talanta, 297, 128630
10. Li Z (2025), ‘Inorganic and hybrid nanomaterials for NIR-II fluorescence imaging-guided therapy of Glioblastoma and perspectives’, Theranostics, 15, 5616
11. Han H (2024), ‘Fabrication of conjugated polymers with aggregation-induced near-infrared-II emission for efficient phototheranostics’, J Mat Chem B, 12, 8383
12. Isuri R K (2025), ‘Clinical Integration of NIR-II Fluorescence Imaging for Cancer Surgery: A Translational Evaluation of Preclinical and Intraoperative Systems’, Cancer, 17, 2676
13. Hong G (2012), ‘Multifunctional in vivo vascular imaging using near-infrared II fluorescence’, Nature Medicine, 18, 1841
14. Hu Z (2020), ‘First-in-human liver-tumour surgery guided by multispectral fluorescence imaging in the visible and near-infrared-I/II windows’, Nature Biomedical Engineering, 4, 259-271
15. Zhang Z (2022), ‘NIR-II nano fluorescence image guided hepatic carcinoma resection on cirrhotic patient’, Photodiagnosis and Photodynamic Therapy, 40, 103098
16. Shi X (2022), ‘Near-Infrared Window II Fluorescence Image-Guided Surgery of High-Grade Gliomas Prolongs the Progression-Free Survival of Patients’, Transactions on Biomedical Engineering, 69, 1889-1900
17. Wu Y (2022), ‘First clinical applications for the NIR-II imaging with ICG in microsurgery’, Cheng Frontiers in Bioengineering and Biotechnology, 10, 1042546
18. Cao C (2021), ‘Intraoperative near-infrared II window fluorescence imaging-assisted nephron-sparing surgery for complete resection of cystic renal masses’, Clinical and Translational Medicine, 11, e604
19. Zhang Z (2025), ‘First-in-human orbital tumor surgery guided by near-infrared II window fluorescence imaging: A feasibility study’, Interdisciplinary Medicine, e20240048
20. Wang F (2025), ‘Guiding esophagectomy with intraoperative NIR-II fluorescence video imaging and rapid computation’, 122, e2517785122

Romain Membrat PhD is a senior R&D leader with extensive expertise in pharma, chemistry and innovative product development. He currently serves as head of the Chemistry Laboratory at Provepharm, where he drives research initiatives focused on formulation and synthesis within pharmaceutical R&D. Prior to this, Romain was responsible for R&D at Ipsomedic, contributing to early-stage development programmes and cross-functional research collaborations. He holds a PhD in chemistry from Aix-Marseille Université and Centrale Méditerranée, France, and his scientific contributions span organic synthesis and chemical methodology. Passionate about translating scientific insight into practical solutions, Romain actively builds research networks and fosters talent within laboratory teams.

Olivier de Beaumont MD MBA is chief development and medical officer at Provepharm, with over 25 years of global experience in clinical development, medical affairs and pharma innovation. He has a strong track record in leading early- and late-stage clinical programmes across internal medicine and oncology, having held senior roles including chief medical officer at NH TherAguix and Onxeo, and leadership positions in drug safety and development at Stallergenes Greer and Aventis. Olivier holds a medical degree and a Master of Business Administration from ESCP, France, complemented by a Master’s in Public Health and Health Economics.