pH-responsive internalisation assays
Seeing what matters: how pH-responsive internalisation assays are reframing MoA in pharma
As targeted modalities proliferate, the ability to quantify ligand, antibody and receptor trafficking into acidic compartments at scale and in near real time is giving teams a far more direct read on drug activity. pH-responsive imaging and sensing approaches are unlocking higher throughput and more physiologically relevant internalisation readouts that bridge discovery, screening and translational workflows, helping to close the long-standing gap between beautiful microscopy and decision-grade data sets
Stéphane Martinez at Revvity
The internalisation inflection point
Drug discovery continues to migrate from broad, non-specific agents towards targeted therapeutics, including receptor-directed biologics, engineered ligands and antibody-drug conjugates (ADCs). In this context, internalisation – the endocytic uptake of cell surface receptors and their bound molecules followed by trafficking through pH-graded endosomes to lysosomes – has become a mechanistic focal point. Measuring whether, how quickly and to what extent a target is ferried into acidic compartments provides direct evidence of on-target engagement and downstream routing that complements affinity, potency and signalling data.1
The challenge has been practical; methods with spatial precision to resolve trafficking often lacked the throughput or robustness needed for screening, while screening-friendly formats could sacrifice relevance or dynamic range. Newer plate-based, pH-responsive assays with built-in background suppression via time-resolved detection are shrinking this actionability gap by translating sophisticated measurements into scalable, workflow integrated tools.
Why pH matters for trafficking readouts
Endocytosis routes cargo through early endosomes (mildly acidic), late endosomes (more acidic) and lysosomes (highly acidic). pH is therefore a natural ‘address label’ for localisation – probes designed to brighten as pH drops can discriminate surface-bound from internalised species and reveal where the cargo is within the endosomal-lysosomal continuum. When combined with time-resolved detection, these assays boost dynamic range and reduce autofluorescence, enabling kinetic and extent of uptake measurements in plates. This is more than a measurement nuance. For ADCs, efficient internalisation into acidic compartments is often a prerequisite for payload release and efficacy. Meanwhile, for G protein-coupled receptors, internalisation can redirect signalling to endosomes, where distinct ‘location-biased’ pathways operate, meaning that measuring trafficking is directly mechanistic, not merely descriptive (Figure 1).2
From artisanal to industrial: the path to high-throughput internalisation
Historically, researchers relied on labour-intensive microscopy workflows such as antibody feeding, pH quenchers and temperature shifts that provide rich context but are hard to scale.
The current wave of pH-responsive, plate-based assays addresses this bottleneck in three ways:
• Acid-activated fluorophores: dyes that remain dim at neutral pH but brighten in endosomes/lysosomes isolate internalised signal from surface background and are available in multiple spectral channels
• Temporal resolution: repeated kinetic reads over minutes to hours enable separation of fast internalisers from slow recyclers, and help map transitions across the endosomal pathway
• Readercompatible, nowash formats: homogeneous assay protocols minimise operatordependent variability and improve reproducibility across plates, days and laboratories. Modern implementations run on widely used multimode readers and standard plate formats, including those compatible with time-resolved fluorescence/time-resolved fluorescence-Förster’s resonance energy transfer instrumentation.
What ‘good’ looks like: four design principles
Physiological relevance without fragility
Assays should tolerate endogenous or modestly overexpressed receptor levels to mirror realistic biology. This is especially important as clinical tissue samples and primary models vary widely. Formats that maintain sensitivity at lower receptor densities help avoid overexpression artifacts and reduce false negatives during hit triage.
High signal-to-background with built-in selectivity
pH-gated signal generation, coupled with time resolved detection, produces strong signal-to-background (S/B) even in complex matrices. This supports confident quantitation of partial internalisers and subtle phenotypes that can be missed in endpoint-only measurements.
Kinetic insight at scale
Capturing full internalisation trajectories reveals distinct mechanismofaction (MoA) signatures, such as rapid endocytosis with lysosomal progression versus slow internalisation with recycling. These kinetic fingerprints can separate candidates that look similar at a single endpoint and inform design decisions.
Seamless instrumentation and analytics
Methods that run on ubiquitous plate readers and integrate with standard data systems lower adoption barriers. Automated curve fitting, trajectory clustering and quality control (QC) checks help convert raw traces into interpretable metrics for ranking, synthetic aperture radar mapping and stage-gate decisions.
Use cases across the pipeline
Early discovery and screening
Internalisation assays can serve as primary or secondary screens for receptor-ligand binding, biologics or conjugates. By ranking both potency and trafficking efficiency, teams can deprioritise binders that fail to internalise or that recycle rapidly without lysosomal delivery, streamlining resources towards candidates with endosomal/lysosomal competence.3
Lead optimisation
Medicinal chemistry and bioengineering decisions such as linker chemistries for conjugates, Fc engineering for antibodies, drug-to-antibody ratio or tag placement within constructs are reflected as measurable shifts in internalisation rates and endolysosomal routing. Multiparameter optimisation, balancing binding, signalling bias and trafficking reduces late-stage surprises and improves translational confidence.4
Translational models
Working at lower receptor expression better reflects clinical heterogeneity. Assays that retain sensitivity under endogenous conditions enable studies in physiologically relevant cell lines or ex vivo systems, supporting biomarker strategies and dose rationales more credibly than overexpression heavy models.
Quality and comparability
Standardised plate-based readouts facilitate cross-site comparisons and longitudinal tracking. When paired with orthogonal measurements (eg, signalling reporters or cytotoxicity), internalisation curves add mechanistic context that can reconcile efficacy and safety observations during candidate selection.
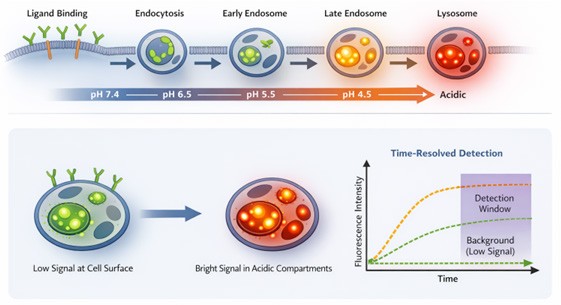
Figure 1: pH-responsive internalisation workflow and detection principle
Practical considerations and pitfalls
Controlling for surface signal
Although pHresponsive assays limit surface interference, they cannot fully eliminate it. To verify that the signal originates from intracellular compartments, include controls such as extracellular quenchers, brief extracellular pH manipulations or gentle proteolytic removal of surfacebound probes. Additionally, assess target specificity by incorporating receptorblocking conditions, genetic perturbations, fluidphase uptake tracers, lowtemperature/energydependent controls and saturation analyses to distinguish receptormediated internalisation from pinocytosis or other nonspecific processes.
Accounting for receptor recycling
Some targets rapidly recycle to the membrane, confounding single time point assessments. Use kinetic sampling and pathway probes, for example, dynasore to acutely inhibit dynamin-dependent internalisation, or bafilomycin A1 to neutralise endosomal/lysosomal pH and test acid dependence of signal accumulation. These tools help deconvolute routes and validate mechanistic assumptions.5,6
Model selection and expression levels
Receptor abundance varies widely between cell systems. Calibrate labelling density and read intervals to maintain sensitivity at lower expression and verify that probe loading does not perturb receptor function or cellular physiology.
Where imaging meets sensing and why spatial analytics matter
Plate based sensing scales discovery, but imaging still provides irreplaceable spatial context, co localisation with compartment markers, visualisation of endosomal maturation, and detection of off pathway trafficking or sequestration. An efficient practice is to rank at scale with homogeneous pH responsive readouts and then validate mechanisms with targeted high content imaging. Recent work also shows that the positioning of receptor containing endosomes can shape transcriptional outputs, underscoring why pairing kinetic sensing with selective imaging yields richer MoA narratives.
Case example
A panel of receptor targeted ligands is tested in cells expressing endogenous receptor levels. A pH responsive internalisation assay provides two hour kinetic traces that resolve three mechanistically distinct phenotypes (Figure 2):
• Fast lysosomal progressors: ligands showing rapid and sustained accumulation in acidic compartments, consistent with efficient trafficking through welldefined clathrinmediated endocytosis into late endosomes and lysosomes – pathways known to support payloadrelease mechanisms such as those used in ADCs
• Recycling biased internalisers: ligands displaying modest uptake with early plateauing, reflecting routing through early endosomes with preferential return to the plasma membrane; a process extensively described for many receptors undergoing clathrin-mediated endocytosis or clathrinindependent internalisation
• Surface-retained binders: ligands demonstrating strong binding in orthogonal assays but minimal signal in acidic compartments, consistent with receptors that internalise poorly, signal weakly or engage pathways (eg, caveolae or clathrinindependent endocytosis) associated with limited acidification.
Selective imaging in a subset confirms co-localisation with early and late endosomal markers, and distinguishes lysosomal routing from recycling pools. Orthogonal signalling assays show that fast progressors reduce surface receptor levels and modulate downstream readouts, aligning internalisation behaviour with pharmacodynamic effects.
Standards, reporting and reproducibility
To enhance comparability across labs and programmes, consider:
• Assay conditions: document pH calibration, temperature, serum conditions, label to target ratios and read intervals
• Core metrics: report initial internalisation rate (slope), time to half max, maximal extent, and recycling indices with fit models and confidence bounds
• Quality thresholds: define minimum S/B and Z′ acceptance criteria at the plate level.
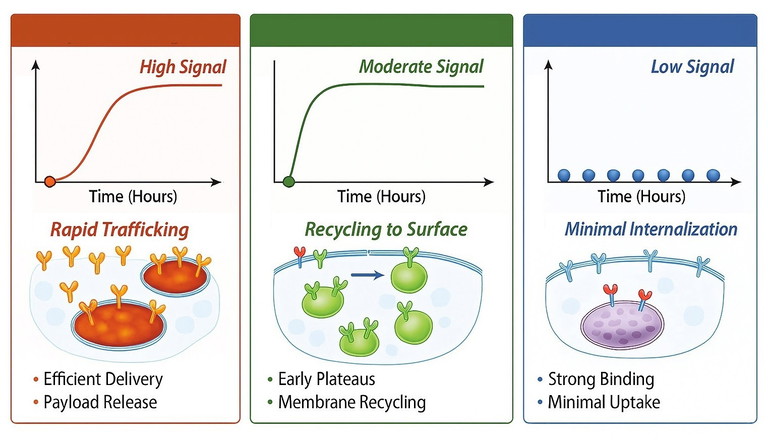
Figure 2: Receptor ligand internalisation assay in endogenous receptor expression
“By integrating kinetic platebased sensing with targeted imaging, applying rigorous QC and designing for analytics from the outset, teams can convert trafficking into a decisionmaking asset rather than an afterthefact explainer”
The road ahead
Deeper multiplexing will pair pHresponsive internalisation with downstream signalling, payload activation, and celldeath or cytotoxicity induction for ADCs within the same plate, enabling teams to link cause and effect and iterate designs more rapidly. Meanwhile, moving into cocultures, primary cells and ex vivo tissues will stresstest internalisation under more physiologically realistic constraints (eg, heterogeneous expression, variable endosomal pH), thereby improving confidence in translational predictions. Finally, adopting an analyticsfirst framework – where intended data outputs and decision criteria (such as ranking ligand performance, clustering phenotypes or generating predictive models) are defined in advance – ensures that experimental design elements like plate layout, temporal sampling and QC parameters are optimised for statistically robust inference. This approach shifts the focus from generating measurements to producing analytically reliable data sets capable of supporting mechanistic interpretation and actionable conclusions. In short, pHresponsive internalisation assays deliver mechanistic clarity at the speed and scale today’s pipelines demand. By integrating kinetic platebased sensing with targeted imaging, applying rigorous QC and designing for analytics from the outset, teams can convert trafficking into a decisionmaking asset rather than an afterthefact explainer.
References:
1. Sorkin A et al (2009), ‘Endocytosis and signalling: intertwining molecular networks’, Nature Reviews Molecular Cell Biology, 10(9), 609-622
2. Flores Espinoza E et al (2024), ‘Beneath the surface: endosomal GPCR signaling’, Trends in Biochemical Sciences, 49, 520-531
3. Khongorzul P et al (2020), ‘Antibody–Drug Conjugates: A Comprehensive Review’, Molecular Cancer Research, 18(1), 3-19
4. Alradwan I A et al (2025), ‘Strategic and Chemical Advances in Antibody–Drug Conjugates’, Pharmaceutics, 17(9), 1164
5. Macia E et al (2006), ‘Dynasore, a Cell-Permeable Inhibitor of Dynamin’, Developmental Cell, 10(6), 839-850
6. Salova A V et al (2025), ‘Deacidification of the Endolysosomal System by the Vesicular Proton Pump V ATPase Inhibitor Bafilomycin A1 Affects EGF Receptor Endocytosis Differently in Endometrial MSC and HeLa Cells’, International Journal of Molecular Sciences, 26(20), 10226

Stéphane Martinez is a seasoned life sciences professional with more than 25 years of experience advancing breakthrough technologies for drug screening and preclinical research. He holds a Master of Science in Engineering in Biochemistry and Immunotechnology, and currently serves as product manager at Revvity, focusing on neurodegenerative diseases and biologics characterisation. His expertise spans fluorescence-based detection, genome engineering and stem cell-based models. With a career covering scientific support, product management, business development and sales, Stéphane brings a uniquely cross-functional perspective and a strong customer-centric approach, building long-term collaborations with leading biotech and pharma innovators worldwide.