SPOT LIGHT
Rethinking early phase development: how to move quickly without creating downstream risk
How important is well-planned early-phase development and the early involvement of contract development and manufacturing organisations in processes?
Marc Lanz and Martin Althaus at CARBOGEN AMCIS
Early-phase development has always required urgency. Today, that urgency is shaped by a more complex set of pressures than ever before. Competitive intensity, milestone-driven financing structures and expanding regulatory expectations are redefining how biotech and virtual pharma companies approach the earliest stages of development.
Dr Marc Lanz, head of Innovation at CARBOGEN AMCIS, believes the industry mindset has shifted noticeably: “We are seeing a move away from simply striving to be best-in-class,” he explains. “Today, many organisations are now focused on being first in class. The commercial value of reaching proof of concept rapidly has increased, and with it the pressure placed on early development programmes.” At the same time, Dr Martin Althaus, manager of process R&D at the company, highlights the impact of changing financial models: “Funding structures have evolved significantly,” he says. “Early-stage companies increasingly operate under staged financing models, in which defined technical milestones must be achieved to unlock further investment. In many cases, scientific delivery is directly linked to financial survival.”
In this environment, early-phase development is not simply about advancing a molecule into clinical evaluation. It is about generating credible scientific data under compressed timelines, while simultaneously laying robust foundations that will withstand scale up, regulatory scrutiny and the realities of commercial manufacturing.
“Speed alone is not enough. It must be efficient, forward-looking and scientifically sound – Marc Lanz”
From speed to efficiency
While speed is critical, CARBOGEN AMCIS deliberately reframes the discussion around efficiency. An exclusive focus on speed risks encouraging what Althaus describes as a “quick and dirty” mindset. Efficiency, by contrast, means using limited time intelligently; delivering material early when required without creating technical debt later in the process. In practice, this means that early supply strategies must be considered alongside long-term manufacturability. Technologies such as simulated moving-bed chromatography may be the fastest way to obtain enantiomerically pure material for initial batches. However, they may not provide the optimal cost structure or scalability at commercial volumes.
Rather than choosing between speed and sustainability, parallel development tracks can be established. While early batches are delivered via the most appropriate route, scalable chemistry and process optimisation can be offered in parallel by the contract development and manufacturing organisation (CDMO). This approach reduces life cycle risk while preserving optionality.
Decisions that persist beyond phase 1
One of the defining characteristics of early-phase development is the longevity of early decisions. Choices made in preclinical and phase 1 – such as around polymorphism, impurity control, salt form selection or synthetic route design – often remain embedded in a programme for years. If these elements are not sufficiently considered, the consequences can be significant. Shifts in impurity profiles may trigger additional toxicology work. Changes in polymorphic form may complicate stability data, regulatory filings or require in vivo pharmacokinetics studies. Bridging toxicology studies and requalification exercises can erode both timelines and budgets. For this reason, CARBOGEN AMCIS emphasises the importance of disciplined early evaluation of key parameters. Polymorph screening, impurity profiling and selection of isolation strategies should not be deferred; they should be integrated early, even when rapid delivery is required. Early development is consequently not a race to generate material at any cost, but a structured process of understanding the molecule and laying out the path for development.
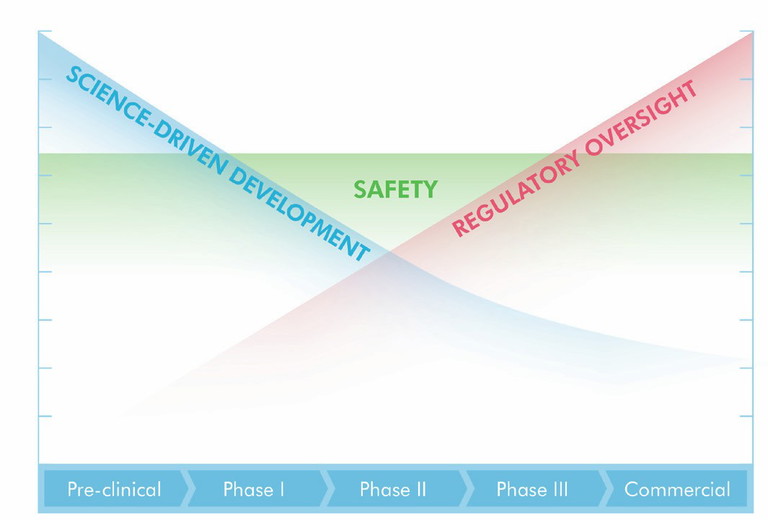
Figure 1: Phase-appropriate continuum. Science-driven early development progressing towards increasing regulatory intensity and commercial scale, with safety maintained throughout
Defining phase-appropriate development
The phrase ‘phase-appropriate development’ is widely used but often misunderstood. For CARBOGEN AMCIS, it is fundamentally about proportionality and timing. In the early stages, development should be science-driven, with scientific understanding, experimental exploration and data generation prioritised. As programmes progress, quality systems, documentation and regulatory controls assume increasing importance. Safety, however, is never variable.
A full safety mindset is embedded from the outset (Figure 1). Calibrating this balance requires experience. For example, large pharma organisations may inadvertently introduce later-stage regulatory burdens too early in the life cycle, slowing progress without adding immediate value. On the flip side, small biotechs may underestimate the importance of documentation and compliance, risking future complications. The role of the CDMO is to moderate and guide, ensuring that development remains scientifically rigorous while avoiding unnecessary front-loading of regulatory requirements. Tools and technologies support this balance.
“Phase-appropriate means knowing when to do what, and when not to over-engineer – Martin Althaus”
For example: AI-assisted analytical method development platforms, such as ChromSwordTM , accelerate column and mobile-phase selection; structured design of experiments enables maximum insight from limited data sets; data integration platforms consolidate analytical information to guide decision-making. However, tools are effective only when applied at the appropriate stage and in alignment with programme maturity, always considering intellectual property protection for the client’s most valuable possession.
Addressing complexity at the right moment
Modern development programmes increasingly involve complex, multi-step pathways and sophisticated impurity challenges. Complexity is no longer the exception; it is the norm. To achieve the programme goals, many elements need to be sequenced strategically, eg, early exploratory trials help eliminate weak options and remove kinks in the route before they become entrenched. While the ultimate decisions are made by the customer, the depth of technical insight provided by an experienced CDMO can significantly shape those choices. Many successful customer relationships are built on this collaborative exchange, where informed discussions guide when to tackle a challenge immediately and when it can be deferred without compromising long-term success. Communication, therefore, becomes central. Transparent discussion of risks, trade-offs and milestone implications strengthens trust and reduces the likelihood of misaligned expectations.
Experience and cross-functional integration
Experience fundamentally alters how early development is approached. Teams that have steered compounds through multiple clinical transitions understand where risks typically emerge and which early decisions influence later scalability. This expertise and accumulated knowledge facilitate forward-looking thinking. Rather than responding to problems as they arise, experienced teams can identify potential bottlenecks before they materialise.
Cross-functional integration is equally vital. During early development, processes are fluid, synthetic routes evolve, analytical methods require adaptation, and impurity signals must be interpreted quickly and addressed correctly. Identifying issues is only the first step; what differentiates effective early-phase execution is the ability to translate those findings into pragmatic solutions and integrate them quickly into the work plan. This requires seamless collaboration across chemistry, analytics, solid-state science, process engineering and safety disciplines.
Equally important is transparency with the customer. Open discussion of challenges, whether anticipated or unexpected, and combined with a proactive, innovative mindset, ensures that adjustments are addressed constructively and without delay. Silos introduce delay, whereas integration enables agility (Figure 2).
“Successful early phase programmes are built on informed dialogue between trusted partners, where technical insight from the CDMO supports confident decisions by the customer – Martin Althaus”
For CARBOGEN AMCIS, partnership is not rhetorical. It reflects a model in which the CDMO acts as an extended workspace by the customer – providing proactive advice, transparency around limitations and risks, and frequent, structured communication to maintain alignment. Regular technical exchanges ensure that challenges are addressed early and decisions remain informed. As CDMOs are increasingly viewed as strategic collaborators rather than transactional suppliers, this partnership mindset becomes a defining differentiator.
Thinking commercially from the beginning
An essential consideration is to think commercially at this early phase. Although cost of goods and environmental impact may appear distant during early development, they become decisive at scale. Solvent selection, isolation efficiency and process throughput directly affect commercial viability at tonne scale. Even if certain adjustments cannot be implemented immediately, early awareness helps ensure clients understand the implications for the future. This transparency strengthens trust and avoids unpleasant surprises at later stages. Keeping the end in mind during the early phase enables smoother transitions and fewer corrective cycles.
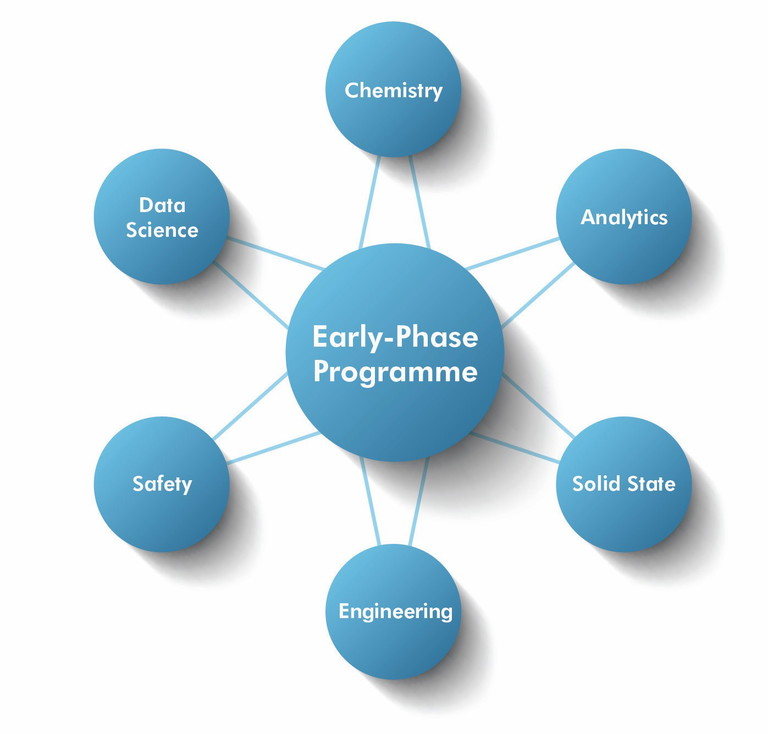
Figure 2: Integrated expertise wheel. Cross-functional expertise in chemistry, analytics, solid state, engineering, safety and data science, integrated around a central early phase programme
The future: insight, agility and structured partnership
Looking ahead, early phase development will rely more on extracting maximum insights from each experiment. Combining integrated analytical data sets, artificial intelligence-supported method development, structured optimisation strategies and knowledge sharing across programmes will speed up learning. However, technical expertise must be balanced with organisational agility. CDMOs need flexible structures that can quickly adapt to different client requirements. Rigid and slow organisational models do not support milestone-driven development environments. In response to this evolving landscape, CARBOGEN AMCIS has formalised its dedicated early-phase offering through SPRINT – a multidisciplinary team designed to support milestone-driven programmes with scientific excellence, regulatory awareness and embedded safety culture. The objective is not simply to move quickly, but to move intelligently, in partnership with clients.
As early phase development continues to evolve, CDMOs that combine efficiency, scientific depth, cross-functional integration and genuine partnership will be best positioned to create lasting value.

Marc Lanz currently serves as head of Innovation at CARBOGEN AMCIS, guiding new technologies and strategic initiatives to strengthen the company’s drug development and manufacturing capabilities. His long tenure at CARBOGEN AMCIS includes progressive roles in Process Research & Development, from project chemist to senior manager, where he contributed to advancing complex chemical process solutions and fostering cross-functional scientific excellence.

Martin Althaus is a chemistry professional with extensive industry experience. Currently manager of PR&D at CARBOGEN AMCIS, he leads scientific and technical initiatives to advance process development for complex chemical active pharmaceutical ingredients, including an interdisciplinary early-development team. Martin began his industrial career at Bachem before joining CARBOGEN AMCIS in 2015, progressing from senior chemist to his current management position.