
Manufacturing
Innovations in pharmaceutical Manufacturing Including Capsuling, Tabletting, Additive Manufacturing, Biomanufacturing Operations

Miriam Krechlok from Mettler-Toledo Product Inspection speaks to IPT about advanced contamination detection systems and how these are utilised in pharma manufacturing to ensure seamless and good manufacturing practice-compliant products

How are life sciences organisations embracing new technologies to help develop soft skills, hopefully mitigating issues seen in the pharma manufacturing sector?

The future of aseptic pharmaceutical manufacturing is gloveless, but what might sound like a bold vision is already turning into reality today

Early-phase development has always required urgency. Today, that urgency is shaped by a more complex set of pressures than ever before. Competitive intensity, milestone-driven financing structures and expanding regulatory expectations are redefining how biotech and virtual pharma companies approach the earliest stages of development.
How is semantic layering technology addressing significant efficiency gaps, creating the infrastructure for more efficient manufacturing operations?
Process innovations provide opportunities to reduce environmental impact while improving efficiency and product quality
How is X-ray CT being used to ensure quality in tabletting?

In biologics development, clone selection is not just a technical milestone, it’s a strategic decision with far-reaching clinical and commercial implications. How can thoughtful clone selection – integrated with developability and manufacturability assessments – accelerate speed to clinic while safeguarding long-term success?

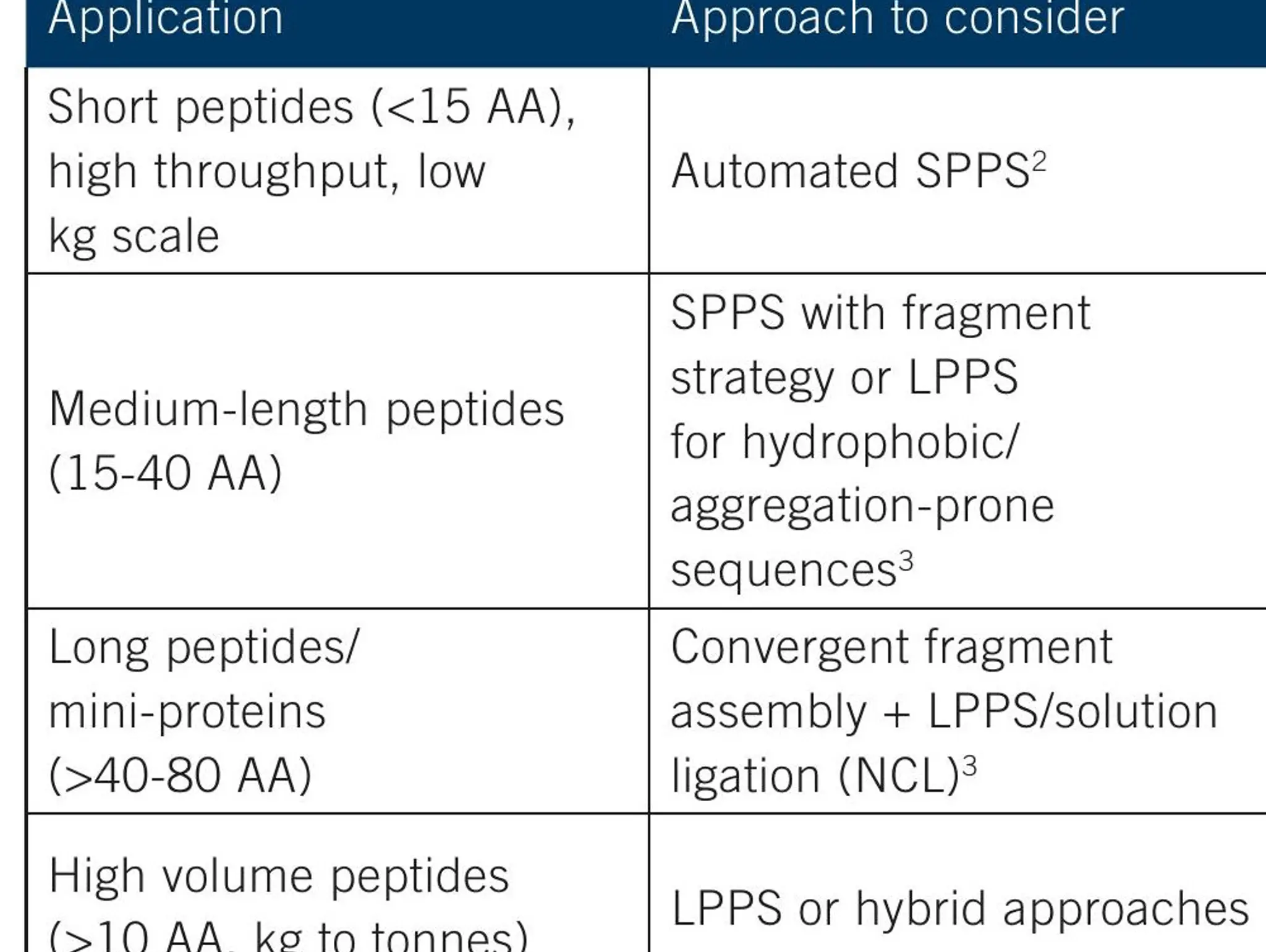
Biocatalysis is enhancing complex molecule process development. By merging the power of enzymes with chemical synthesis, scientists and manufacturers are addressing long-standing challenges in stereoselectivity, route complexity and sustainability

SPOTLIGHT: How CARBOGEN AMCIS supports complex molecules and challenging formulations from early development to commercialisation

What are eight compelling reasons why manufacturers should prioritise the use of advanced contamination detection systems in their operations?

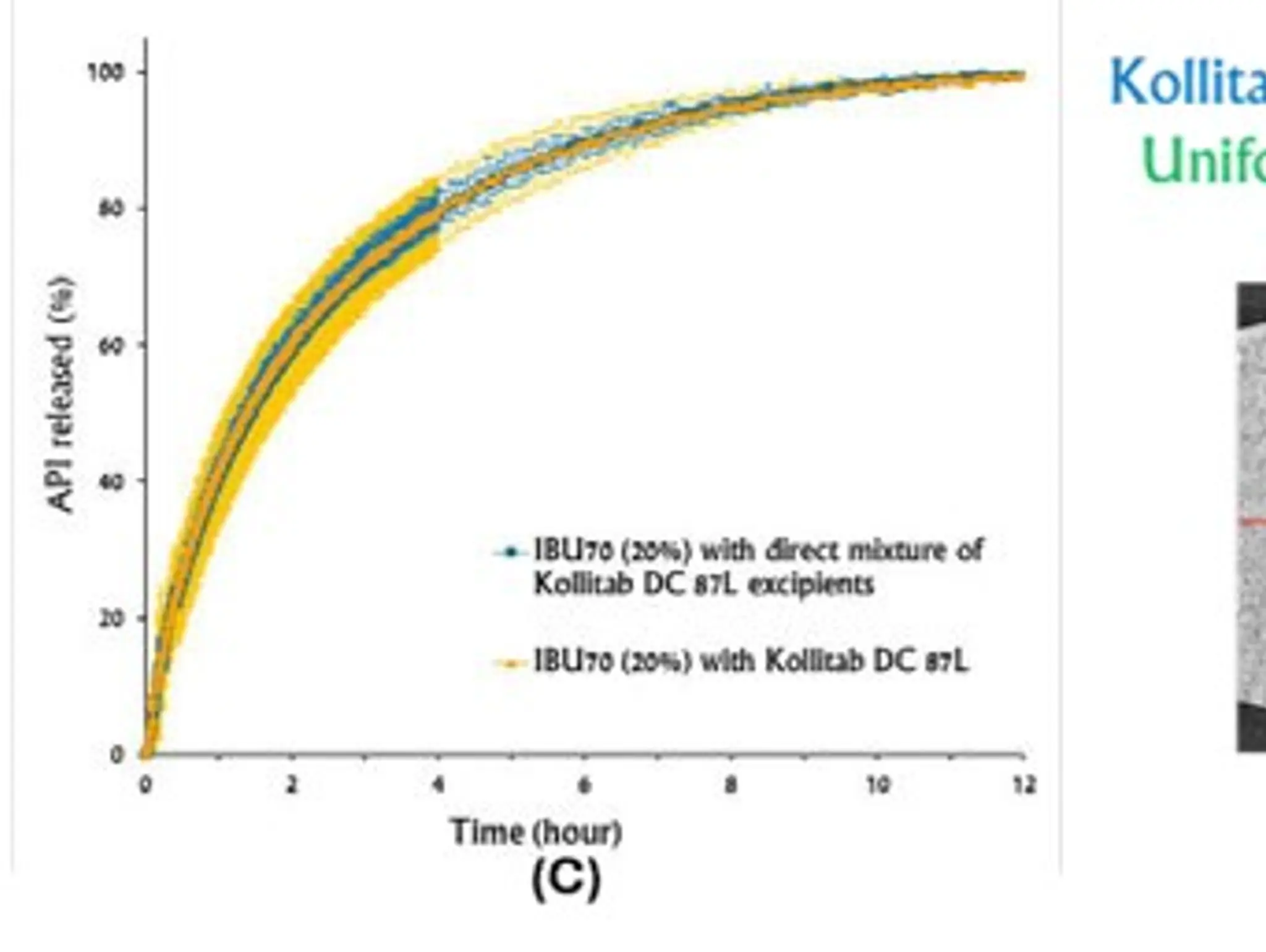
SPOTLIGHT: Bill Supplee at Natoli Engineering discusses the best ways to ensure tablet density uniformity in pharma products